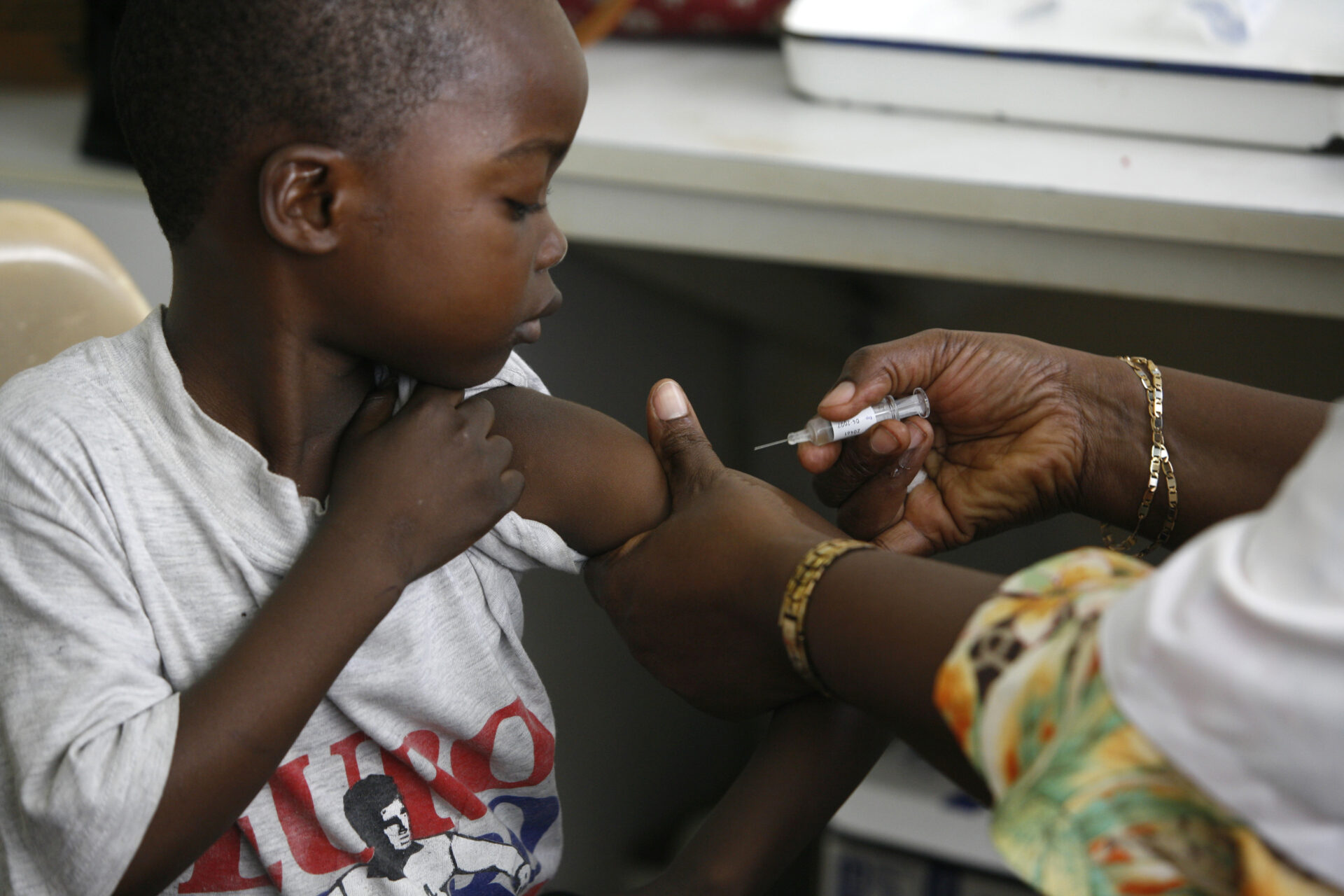
Access to medicines is fundamental to good health. Yet over two billion people – nearly one-third of humanity – continue to lack access to essential healthcare supplies.
Governments working to keep their people healthy face many challenges, but avoidable delays in getting critical medical supplies across borders should not be among them. Slow inspections, lack of facilities, and paper-based procedures can often deprive those in desperate need.
This doesn’t have to happen. Trade facilitation cannot overcome every healthcare challenge, but it can contribute by identifying and unblocking border logjams, ensuring that critical health supplies reach their intended destinations faster and more cost-effectively.
Streamlining processes can speed up delivery while helping regulatory authorities to protect people from falsified products.
Trade facilitation cuts red tape and improves clearance procedures, ensuring that all legitimate goods, including healthcare supplies, clear borders as efficiently as possible. Border reforms can strengthen medical supply chains, improve administrative efficiency, and contribute to building more robust healthcare systems.